Biogen, Eisai Surge as Alzheimer's Drug Shows Promise in Trial
This article for Bloomberg may be of interest to subscribers. Here is a section:
“There is an important cautionary note however: the magnitude of the delay – which was a slowing of decline – was small,” he said. “We can only hope that the benefit is durable and could grow with time. Those long-term properties are unknowable at this time.”
The medicine was originally licensed from Sweden’s BioArctic AB, whose shares more than doubled on the news.
The Alzheimer’s Association welcomed the results, saying they were the most encouraging findings to date from drugs aimed at treating the underlying causes of the disease. Lecanemab has the potential to change the course of the disease and help people in the earliest stages retain their abilities, remain independent and fully participate in daily life, the group said.
Pharmaceutical and biotechnology analysts were equally bullish. “We finally have what we believe to be a clean win in Alzheimer’s disease,” Evan David Seigerman, an analyst at BMO Capital Markets, wrote in a note to clients. “The top-line data are clear to us — lecanemab slows the rate of cognitive decline.”
The trial met every goal that was set, including other measures of mental function and the ability to perform daily activities, the companies said.
The biotechnology/pharmaceuticals sector is endlessly productive. It is one of the few where there is genuine potential for new products to meet unmet needs. That means there is clear scope for value creation that can contribute to both better living standards and rising productivity.
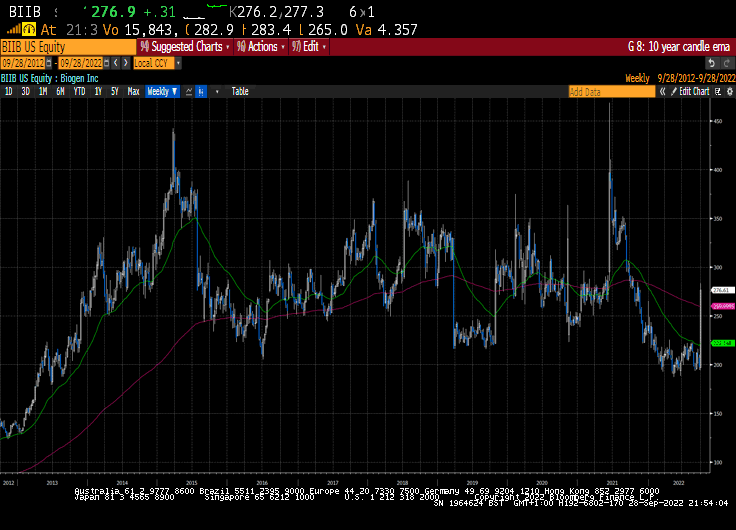 Alzheimer’s and arthritis are the two big chronic conditions that did not have prescribable solutions. Biogen has been yoyoing around based on expectations of whether its Alzheimer’s drugs are effective. The share rebounded emphatically from the lower side of its seven-year range today.
Alzheimer’s and arthritis are the two big chronic conditions that did not have prescribable solutions. Biogen has been yoyoing around based on expectations of whether its Alzheimer’s drugs are effective. The share rebounded emphatically from the lower side of its seven-year range today.
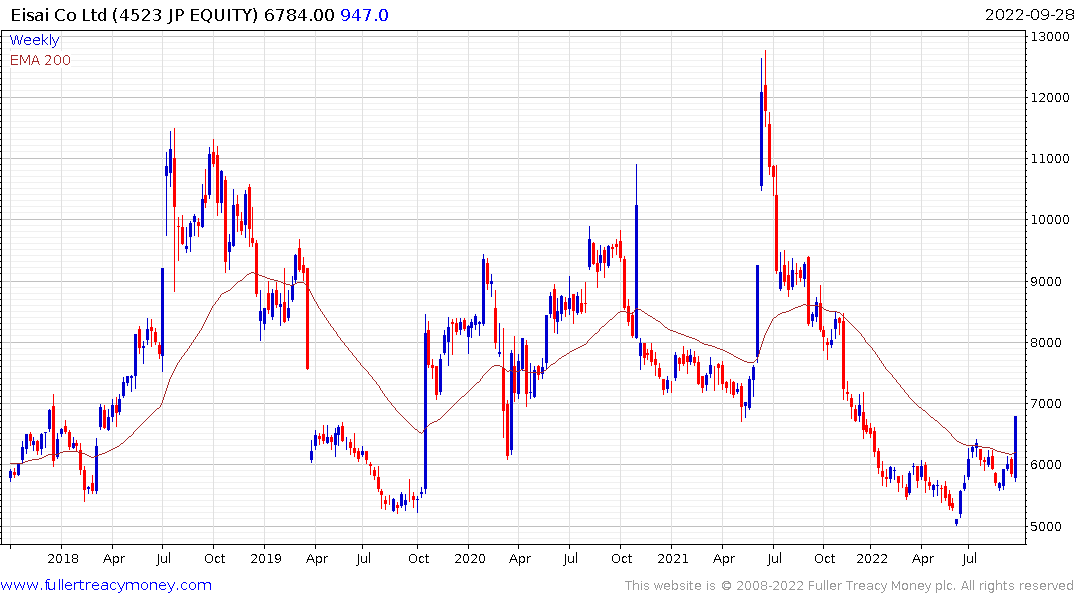 Eisai has a similar pattern.
Eisai has a similar pattern.
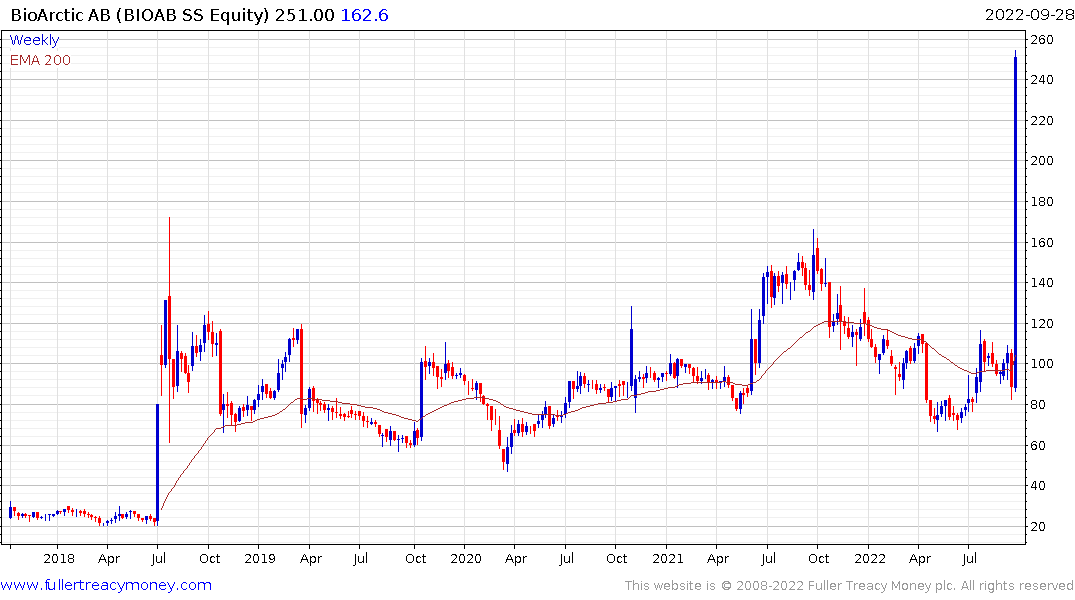
This news was truly transformative for BioArtic as it broke out of a four-year range.


